
MedTech firm reaches for the stars
By Stephen Law, Editor EP&T
Electronics Medical designs designs Electronics electronics medical medicalStarFish Medical specializes in designing medical and life science devices

Starfish Medical has operated as a full service medical device designer and contract manufacturer in Victoria BC and Toronto for more than 20-years.
Referring to his firm as a “pre-eminent” medical-device design company in Canada, founder and CEO of Starfish Medical Scott Phillips has reason to be proud and assured. Based in Victoria BC and an office in Toronto, StarFish has grown under his leadership into a diverse professional organization with clients around the world and 100% focus on medical devices.
Supporting his firm’s credibility within medical circles is the number of industry awards and recognition it has received. Some accolades include: The Globe and Mail’s ranking of Canada’s Top Growing Companies in 2019; and Growth 500 ranking for the second consecutive year, BC Export Leadership Award, Strategic Life Sciences Partner of the Year in 2018. In 2017 Phillips captured the EY Entrepreneur of the Year and was a Pacific Awards Technology Category Winner, while landing a Medical Design Excellence (MDEA) Award.
For the past 20-years StarFish Medical has operated as a full service medical device design CEM offering turn-key design, development, and manufacturing services. The firm uses its Pathfinder process to reduce wasted effort and increase success for medical device product definition, technical engineering, and product development. Prototype and volume production are delivered within an ISO 13485 certified Quality Management System and an FDA registered manufacturing and clean room facility.
EP&T posed a number of questions to Phillips, who holds a degree in engineering physics from UBC. Prior to starting StarFish, he worked in diverse areas such as lithium battery development and manufacturing, UV spectroscopy instrumentation and hi-fi audio speakers.
——————–
How did you create StarFish?
I formed StarFish Medical in Victoria in 1999 as an outgrowth of my own contracting activities. I was 10 years into my career at the time as an engineering physics grad from the University of BC. Now we employ 140 people, mostly in Victoria and Toronto. We serve the North American market and are one of the leading companies in our space. StarFish focuses 100% on medical devices and life sciences products – primarily complex electromechanical design, engineering and manufacturing services. We also have expertise in optics, cell therapies, and microfluidic assays.

Starfish Medical founder and CEO Scott Phillips holds a degree in engineering physics from UBC.
Detail the Pathfinder process, used to reduce wasted effort and increase success for medical device product definition, technical engineering, and product development.
Pathfinder is our framework for product definition. We really try to engineer an optimal business outcome for our clients. We make sure we’re properly addressing issues including usability, regulatory compliance, target margins, consumables strategy, clinical strategy, IP, reimbursement and service strategy. The investigation is framed into formalized design gates to be as efficient as possible at designing and engineering the right product.
What challenges are medical electronic designers faced with, that other consumer-based electronic design houses never have to.
Medical electronic designers have a lot of interesting regulatory issues to deal with from government bodies like Health Canada, FDA, and EU. For example, meeting industry specific standards such as ISO13485, IEC60601 (safety of electronic medical devices), IEC62304 (software for medical devices), ISO 14971 (risk management for medical devices), ISO62366 (usability for medical devices). Formative and summative human factors studies are required. Change control is a big deal. Often changes have regulatory implications that require careful attention. The Design History File of a device must include updates to the risk analysis as changes occur and field data comes back. The lifetime of a medical device can be a decade, so we have to plan on how to support devices in the face of component obsolescence.
What was StarFish’s initial focus or area of expertise within the medical electronics design sphere. How does that compare to what it does today?

Most Starfish staff worked remotely through the pandemic, while a handfull came in to test benchtop prototypes.
21 years ago we started in ophthalmic instrumentation. Our first large program in medical was an ophthalmic ultrasound system. Today, StarFish assisted patents span over 150 cardiovascular, digital health, IVD, ophthalmology, optics, and ultrasound medical devices. Examples include 3D ultrasound from inside the beating heart, spinal surgery for ALS patients, and platelet analysis using lasers for safer transfusions.
Outline your involvement with the Next Generation Manufacturing Supercluster, in helping to design a ventilator model.
We were contacted in late March by NGen (the Next Generation Manufacturing Supercluster). They connected us with Cerebra Health, a group headed by Dr. Magdy Younes in Winnipeg that had old clinical prototypes and wanted to adapt them to the current circumstances. Dr. Younes is one of the world’s leading ventilator innovators. In very short order we created a collaboration and started working on the design. Within a week we were able to deploy over 25 engineers and designers onto the program. NGen provided initial funding through their COVID-19 program, and the Federal Government placed a large order. It’s an honour to have the opportunity to step up at this critical time.
We’re currently in the design cycle with supply chain integration happening in parallel. Early production is expected in May. Clinical and regulatory discussions are also occurring at the same time. You can imagine the range of challenges we’re working through.
How many units will be produced? Where will they be distributed?
As announced by Prime Minister Trudeau, the government intends to buy 30,000 made in Canada ventilators produced by four groups. The Winnipeg Ventilator is one of the four. Our supply chain group is finalizing the CM partners shortly. With an even division of production, we’ll be responsible for 7500. But that may go up or down depending on the other parties and the evolving need.
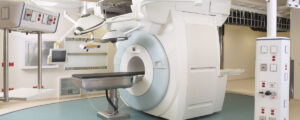
IMotion Neuro MRI Family of products designed by Starfish Medical.
How is StarFish dealing with the restrictive capacity within supply chains during the COVID era?
Our supply chain group is working extra hard. We have been successful with securing parts for the other products in our plant and are working to obtain the right parts for our ventilators. Luckily we can draw on the supply chain capacity and influence of other larger partners as well.
Has StarFish experienced other challenges amid the disruption caused by this pandemic?
It’s too early to tell. It’s certainly harder to visit our clients. Clinical testing is largely on hold for the moment as well. In the short term we’ve seen more emphasis on programs that are somehow aligned with Covid-19.
Is most of your staff (engineering, designers) working remotely during the COVID-19 lockdown? If so, how is the team remaining connected, focused and collaborating on existing designs?

IOPac portable opthalimic device.
Most of our engineering and design staff is working from home, with only a few people coming into the office to test benchtop prototypes and the like. Collaboration is happening mostly on-line with video tools and other document sharing platforms. We’ve added more communication tools and scheduled huddles to replace ad-hoc conversations. Most deadlines are being met, however, some supply chain bottlenecks beyond our control are affecting shipments a little.
Clearly StarFish specializes in complex design prototyping. How do you handle production runs? Do you use a local CEM?
We are a contract product developer. Often we have a big impact on the requirements development and strategic market fit of the products we develop as well. As such we often create a variety of prototypes for different purposes. Our manufacturing team focuses on NPI and clinical needs as well low volume, complex medical devices. We outsource electronics assembly and have a range of different CEMs we use for different volume requirements. Some CEMs are as far away as China, and others within a few blocks as we balance speed and cost.
